Ammonia (NH3) synthesis from nitrogen (N2) and hydrogen (H2) is an important chemical reactions. Because of the high stability of the N≡N triple bond in N2, industrial NH3 production mainly relies on the Haber-Bosch process, which converts N2 and H2 to NH3 under high temperatures (350 - 500 oC) and high pressures (10 - 30 MPa). This process is highly energy-intensive and associated with substantial carbon emissions.
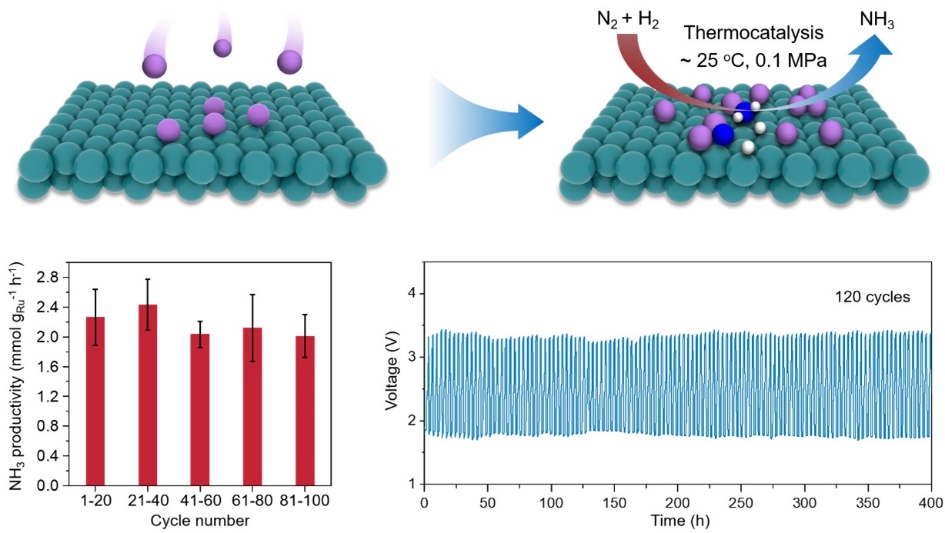
Schematic illustration and the performance of the thermocatalytic ambient-condition ammonia synthesis from N2 and H2 at the metallic Li/Ru interfaces (Image by HOU Ying)
In a study published in Chem, a research team led by Prof. DENG Dehui and Prof. YU Liang from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS), along with Prof. CUI Yi from the Suzhou Institute of Nano-Tech and Nano-Bionics of CAS, developed a novel catalyst which enables thermocatalytic conversion of N2 and H2 to NH3 at room temperature and ambient pressure.
The researchers deposited metallic lithium (Li) on ruthenium (Ru) surface to form highly active Li/Ru interfaces. They found that this interface exhibited a synergetic effect that promoted both N2 activation and hydrogenation steps. Electron donation from Li to the antibonding orbital of adsorbed N2 facilitated N2 dissociation, while Li-N bonding interaction favored the hydrogenation of NHx intermediates.
Moverover, the researchers demonstrated the ambient-condition NH3 synthesis in a reversible Li battery (RLB), with Li metal as the anode and well-dispersed Ru nanoparticles on carbon nanotubes (Ru/CNTs) as the cathode. They found that Li/Ru interfaces were generated in situ during battery discharge. With a mixture of N2 and H2 flowing across the Li/Ru interface on the cathode, an NH3 productivity of 2.43 mmolNH3 gRu-1 h-1 was achieved at about 25 oC and 0.1 MPa.
Furthermore, the charge-discharge cycling of the RLB enable the in situ generation and regeneration of the Li/Ru interfaces. This process operated stably for over 400 hours across more than 120 cycles.
"Integrated with high-efficiency energy-storage RLB systems, this process provides a new way to establish a low-energy and sustainable paradigm for NH3 synthesis," said Prof. DENG.